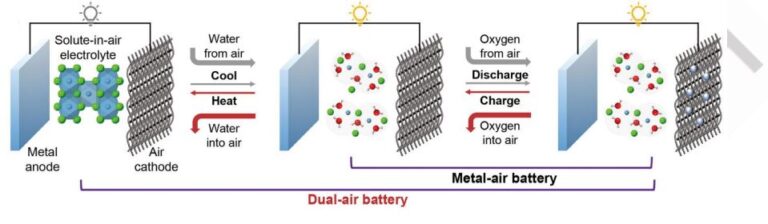
Thermal safety issues of batteries have hindered their large-scale applications. Non-flammable electrolytes improved safety but solvent evaporation above 100 ºC limited thermal tolerance, lacking reliability. Herein, fire-tolerant metal-air batteries were realized by introducing solute-in-air electrolytes whose hygroscopic solutes could spontaneously reabsorb the evaporated water solvent. Using Zn/CaCl2-in-air/carbon batteries as a proof-of-concept, they failed upon burning at 631.8 ºC but self-recovered then by reabsorbing water from the air at room temperature. Different from conventional aqueous electrolytes whose irreversible thermal transformation is determined by the boiling points of solvents, solute-in-air electrolytes make this transformation determined by the much higher decomposition temperature of solutes. It was found that stronger intramolecular bonds instead of intermolecular (van der Waals) interactions were strongly correlated to ultra-high tolerance temperatures of our solute-in-air electrolytes, inspiring a concept of non-van der Waals electrolytes. Our study would improve the understanding of the thermal properties of electrolytes, guide the design of solute-in-air electrolytes, and enhance battery safety.
Researcher/Author:
Huarong Xia, Shengkai Cao, Zhisheng Lv, Jiaqi Wei, Song Yuan, Xue Feng, and Xiaodong Chen
Published in: Angew. Chem. Int. Ed. 2024, e202318369
To download the paper, please proceed to:
https://doi.org/10.1002/anie.202318369

